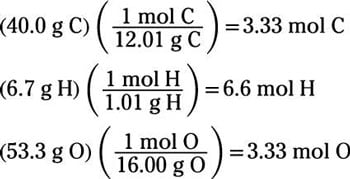Unbelievable Tips About How To Write Empirical Formula

Spq‑2 (eu) , spq‑2.a (lo) , spq‑2.a.3 (ek) transcript.
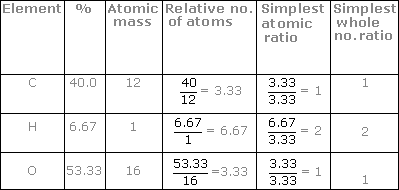
How to write empirical formula. 5) compute the empirical formula weight: 32 + 16 + 16 = 64. Calculate the mass of each element in grams. In the phosphorus oxide example the lowest number is 4.
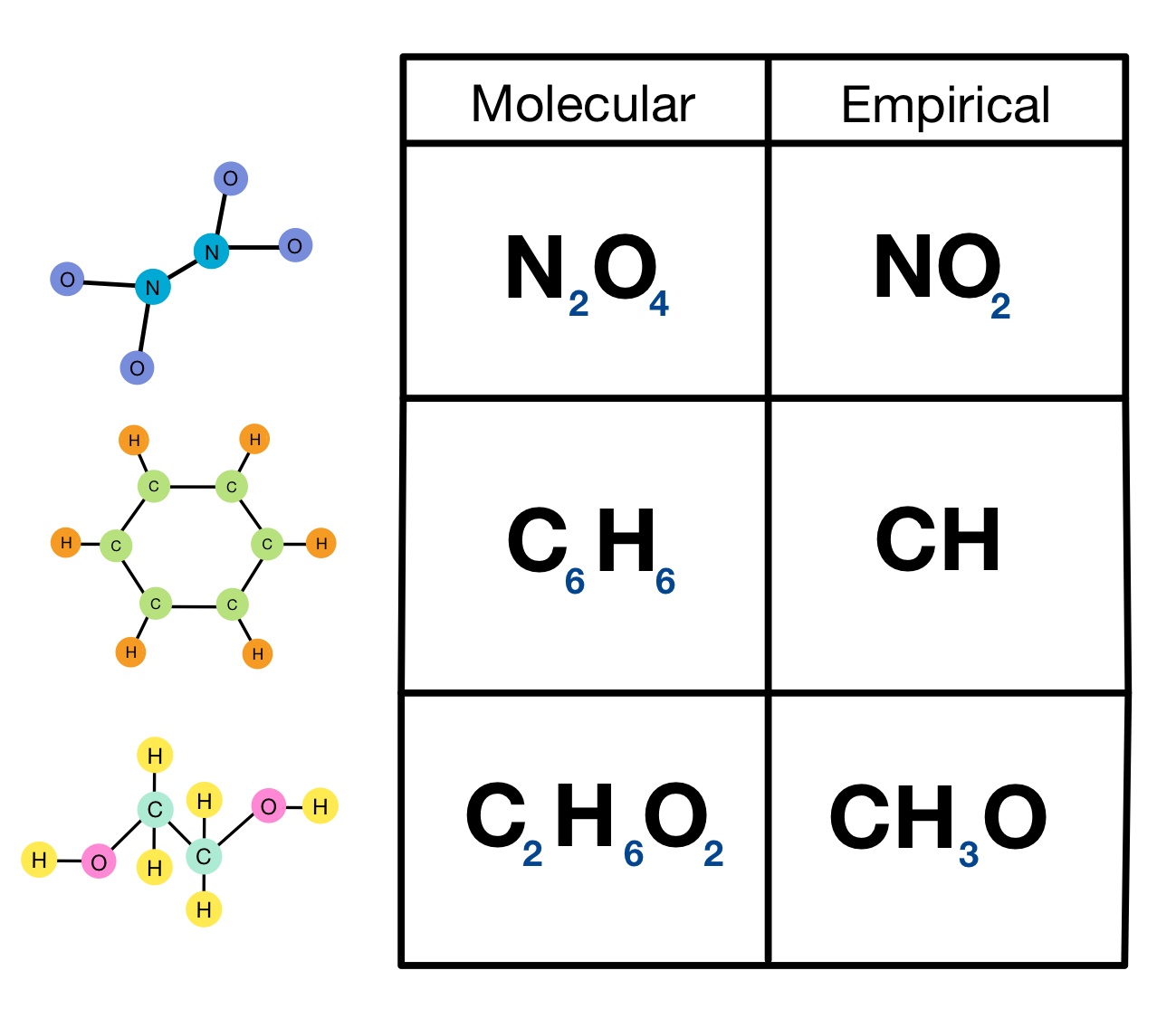
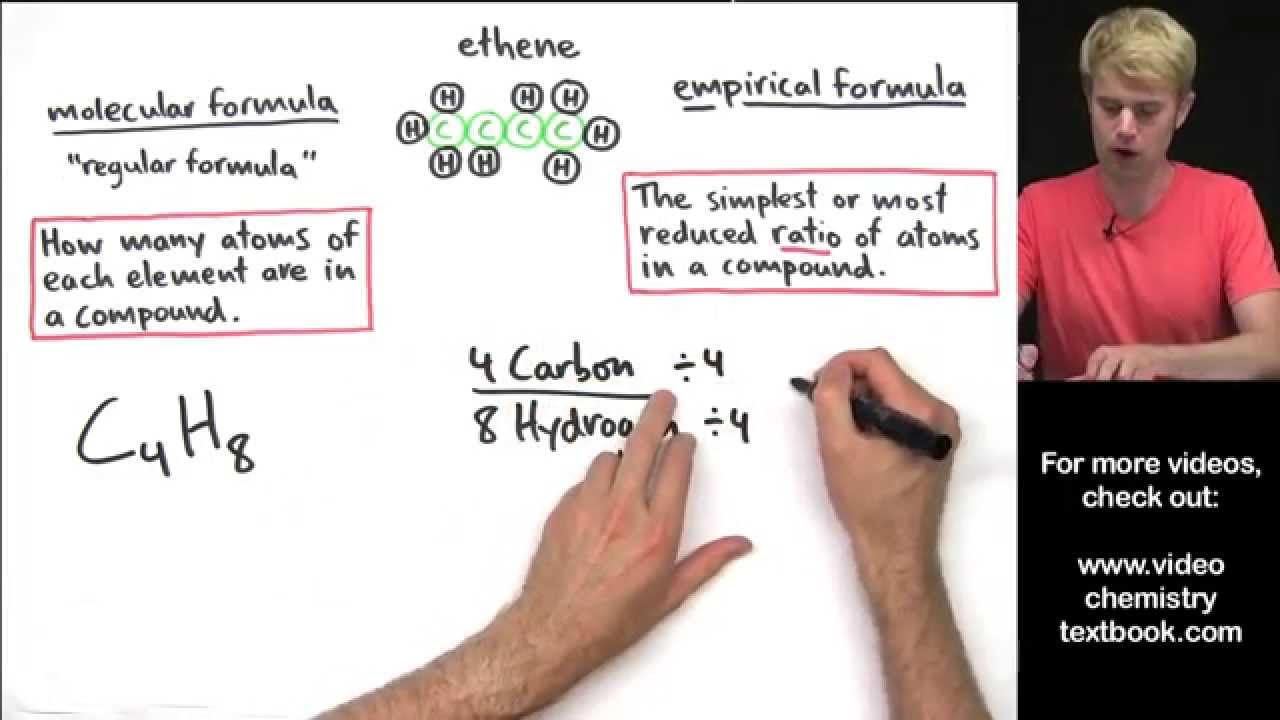
Multiply all the subscripts in the empirical. For example, the empirical formula of a hydrocarbon is ch2 and its mr is 42. The empirical formula of a chemical compound is a representation of the simplest whole number ratio between the elements comprising the compound.
The result should be a whole number or very close to a whole number. How do you write an empirical formula? This video goes into detailed steps on how to find the empirical formula of a compound.
Professional authors can write an essay in 3 hours, if there is a certain volume, but it must be borne in mind that with such a service the price will be the highest. Divide the molar mass of the compound by the empirical formula mass. The mass of the atoms in the empirical.
Order the elements according to the. 4) write the empirical formula: There are three main types of chemical formulas:
Hooray for no more confusion!check out my new complete guide on empir. Add up the atomic masses of the atoms in the empirical formula. The steps for determining a compound’s empirical formula are as follows:.
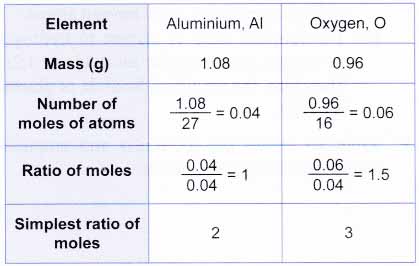
Find the ratio or the moles of each element by dividing the number of moles of each by the smallest number of moles. You can discover the empirical formula by counting the number of each atom in a compound and dividing it by the lowest number. 6) divide the molecule weight by the efw: 64.07 / 64 = 1.
If we know which elements are present in a molecule and in what ratio, we can. Such essays are very difficult to write, because many are not interested in this and do not see the meaning of the text. Here is an example of determining empirical formula of an ionic compound.
How to calculate empirical formula? 7) use the scaling factor.